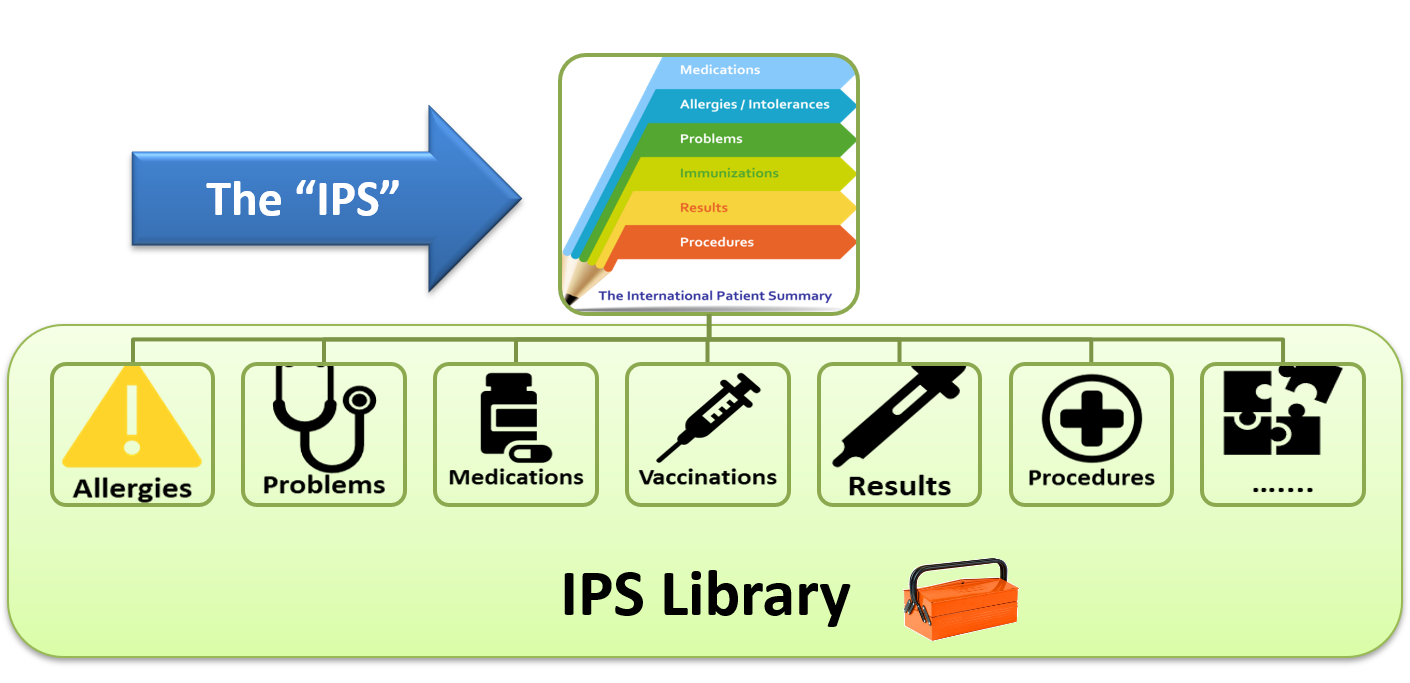
The International Patient Summary (IPS) is a key standard for global interoperability. Its goal is to create a common foundation for patient summary exchange across borders and ensure the ability to share key information with caregivers whenever and wherever needed. Several nations and international organizations have selected the IPS as a baseline for their summaries, and recently major electronic health record (EHR) and health information technology (IT) vendors have shared their planned implementations. These include Epic, MEDITECH, CommonHealth, Google, Patient Centric Solutions, VeroSource and Verto Health. Like many successful standards, today’s progress has been years in the making.
EHRs and Health IT Vendors Accelerate IPS Adoption
[fa icon="calendar'] Aug 1, 2024 1:00:00 PM / by John D'Amore, MS posted in CDA, IHE, International Patient Summary, FHIR Connectathon, FHIR Implementation Guides, IPS, EHR integration
Advancing the International Patient Summary
[fa icon="calendar'] Dec 1, 2021 10:39:47 AM / by HL7 posted in FHIR, CDA, HL7, HL7 community, health IT, International Patient Summary, FHIR Community, IPS, International Patient Access, IPA
Spring 2021 CTO Tooling Update
[fa icon="calendar'] May 12, 2021 11:07:30 AM / by Wayne Kubick posted in FHIR, CDA, HL7, health IT, C-CDA, news, tooling, JIRA, Confluence, publishing, UTG, FHIR registry
Déjà Vu All Over Again
My last tooling update was titled Focus on Finishing. Thus, in homage to the inimitable Yogi Berra, it would be hypocrisy to change focus now. Focus on Finishing is still the principal theme for the year, building on essentialism, my other guiding light, as expressed in the axiom “Do less, better.”
Toward that end, we continue to move ahead with our transition to our core collaboration tool stack and processes based on workflow-driven online forms. As of this writing, we’re completing final improvements to make the online PSS available to all later this spring. We’ll be working to finish automating most other key form-driven processes after that.
In addition, we hope to finish our transition to a new JIRA-based balloting system, which is also being piloted as of this writing. This, together with the recent transition from GForge Tracker and the STU Feedback web page to JIRA, puts all of our specification feedback in one repository moving forward.
While finishing our transition for balloting is critically important, we also have to update and replace some peripheral systems supporting the balloting process for members, as well as our core business systems for managing membership, events and operations. While we don’t expect to complete this transition to a new Association Management System before the end of 2021, we’ll be focused on finishing this as rapidly as possible since it’s an essential foundation to further systems improvements for the HL7 organization.
C-CDA Implementation-A-Thon to Expand Outreach to Engage New Communities
[fa icon="calendar'] Mar 1, 2021 3:53:46 PM / by Lisa R. Nelson, MS, MBA posted in CDA, HL7, HL7 community, health IT, C-CDA
The CDA Management Group (CMG) aims to use the next C-CDA Implementation-A-Thon (IAT) to expand outreach to engage new communities and increase the impact of this content improvement effort. “We learned so much last year about how to maximize the value of implementer-led implementation-a-thons,” said CMG Co-Chair Lisa Nelson. “The new approach was a step in the right direction. It helped implementers drive the conversation and focus the community on making changes that would yield valuable improvements,” she explained.
New HL7 C-CDA Navigation Tool Released
[fa icon="calendar'] Feb 23, 2021 10:12:20 AM / by John D'Amore, MS posted in CDA, HL7 community, interoperability, C-CDA, tooling, implementation, implementation guide
Primary author: John D'Amore, Co-Founder, Diameter Health; Co-authors: Brett Marquard, Principal, Wave One Associates and Wayne Kubick, CTO, HL7 International
While HL7 FHIR® (Fast Healthcare Interoperability Resources) is today’s hottest healthcare standard, clinical documents are already exchanged in the billions today. HL7 published the Consolidated Clinical Document Architecture (C-CDA) in 2011 to support care coordination and patient engagement. The ONC 2014 certification rule named C-CDA R1.1 and adoption exploded. The current version of C-CDA, R2.1, remains backwards compatible to that version today. While C-CDA is a flexible, robust standard to record patient care longitudinally or for an encounter, it is structurally complex. The C-CDA standard itself is over 1,000 pages long. Applying the standard to the latest US requirements for clinical data exchange, known as the US Core Data for Interoperability, also routinely requires information from the C-CDA Companion Guide.
HL7 publishes the C-CDA standard and its Companion Guide as PDF documents. That will remain the official version of the standard, but a small team from the CDA Management and Structured Documents Working Groups saw an opportunity to make the C-CDA content easier to use by developing a new web tool. Since the C-CDA has reusable parts, known as “templates,” it seemed logical to make each template searchable and distinct via unique web pages so the standard would be much easier to absorb.
The new HL7 C-CDA Online Search Tool is a searchable, web-based navigation resource for the Consolidated Clinical Document Architecture (C-CDA 2.1) and its Companion Guide. The solution includes over 240 unique web pages that make each template accessible on the web with linkage back to the original PDF content. This navigation is managed through a single searchable index page, like a smart table of contents.
Using the template webpages, health IT vendors can ensure that their C-CDAs are conformant and high-quality for document exchange. The search tool enables users to search the C-CDA by description, template object identifier (OID) or conformance number. Conformance number searches are particularly valuable for C-CDA implementers, since validators will return these numbers when there’s a violation or warning associated with C-CDA testing.
What is HL7 + Introduction to Product Lines
[fa icon="calendar'] Dec 3, 2019 12:30:08 PM / by Carol Macumber, MS, PMP, FAMIA posted in FHIR, CDA, Version 2, interoperability, C-CDA, Version 3, CMS, Health Quality Measures Format, Quality Reporting, Clinical Quality Language
WHY STANDARDS?
C-CDA Implementation-A-Thon in September 2019 as Part of HL7 FHIR Connectathon
[fa icon="calendar'] Jun 20, 2019 11:32:39 AM / by Dave Hamill posted in CDA, HL7 working group meeting, C-CDA, Connectathon
The next Consolidated Clinical Document Architecture (C-CDA) Implementation-A-Thon (IAT) is scheduled this September 14-15 in Atlanta, Georgia. It will be held as a track within the HL7 FHIR Connectathon, allowing attendees to optimize participation across CDA and FHIR-related tracks.
How to Get HL7 Certified
[fa icon="calendar'] May 13, 2019 1:51:08 PM / by Sadhana Alangar, PhD posted in FHIR, HL7 education, CDA, HL7, certification, Version 2, Version 3
Did you know HL7 offers certification and proficiency exams for its healthcare information technology standards?
CTO Tooling Update: Neither a Sprint nor a Marathon
[fa icon="calendar'] Apr 29, 2019 3:42:48 PM / by Wayne Kubick posted in FHIR, CDA, HL7, health IT, C-CDA, news, tooling, JIRA, Confluence
Our ongoing tooling journey at HL7 continues, neither as a sprint nor a marathon. For us, it’s really more like an odyssey – an ongoing journey where there is always something more to be done, another path to explore, and a final destination (retirement, for example) seems far out of reach. In the case of HL7 tooling, a fair number of tooling retirements are well overdue.
Despite the wait, it’s gratifying to see when tangible progress is actually achieved. On the Confluence front, we’re in the home stretch of phase 1 of the rollout, though there’s a whole new course to pursue just around the bend. We now have all work groups on Confluence (!) and have also migrated many more projects, committees and collaborations. New functions and help features in Confluence (including a major facelift for confluence.hl7.org) are being added regularly, and you can keep up with these by checking the CTO Tooling Update page. This enabling platform is already unleashing many new opportunities within the HL7 community. Our next target is to work toward optimizing our processes with online forms and workflow. The online project scope statement (PSS) pilot is now available and will give us an opportunity to speed up reviews and approvals as well as make new projects more visible to the community in the hope we can avoid last minute catchups.
5 Ways to Market Yourself With HL7 Certification
[fa icon="calendar'] Aug 21, 2018 4:37:40 PM / by Sadhana Alangar, PhD posted in FHIR, HL7 education, CDA, certification, Version 2, Version 3
Are you HL7 certified?
HL7 offers certification in its primary standards for health information technology including Version 2 (V2), Version 3 (V3), Clinical Document Architecture (CDA®) and HL7 Fast Healthcare Interoperability Resources (FHIR®). The testing is designed to help professionals achieve industry-recognized levels of proficiency and expertise.